B-lymphoblastic acute leukemia (B-ALL) is the most prevalent cancer among children, and prophylactic central nervous system (CNS) therapy has significantly contributed to high cure rates for the past 50 years. However, relapses in bone marrow and/or CNS continue to pose challenges to achieving cure for all patients, especially in those with high-risk disease. CNS status at diagnosis, defined as CNS1 (no blasts present), CNS2 (blasts with <5 total white blood cells [WBC] per mm 3 in a cerebrospinal fluid [CSF] sample), and CNS3 (blasts with ≥5 total WBC per mm 3 or grossly traumatic), plays a crucial role in determining treatment stratification. In the past, patients with CNS1 and CNS2 received similar therapy, but evidence suggested that those with CNS2 were at a higher risk for marrow and/or CNS relapse. As a result, the Children's Oncology Group (COG) frontline trials AALL1131 and AALL0932 implemented additional CNS therapy for CNS2 patients beginning in 2012 and 2016, respectively, specifically twice weekly intrathecal chemotherapy until CSF cleared of disease 3 consecutive times. This change highlighted the necessity for accurate identification of CNS2. Based on anecdotal observation, we hypothesized that the incidence of CNS2 among pediatric B-ALL patients at diagnosis was higher after this therapeutic change due to pathologist awareness of its implications for patient management.
The electronic medical records of all consecutive pediatric patients diagnosed with B-ALL at Children's Hospital Los Angeles (CHLA) from September 2008 to December 2019 were reviewed. CSF WBC count and blast involvement were used to calculate CNS status. Blast involvement was determined by one or more pathologists per case by morphologic review of cytocentrifuged CSF material. The patients were divided into two cohorts, one before (cohort A) and one after (cohort B) the COG AALL0932 protocol change (May 20, 2016). Differences between cohorts were evaluated via Fisher's exact test and Wilcoxon rank sum test. All tests were two-sided, except for CNS2 status, and significance level was set a priori at 0.05.
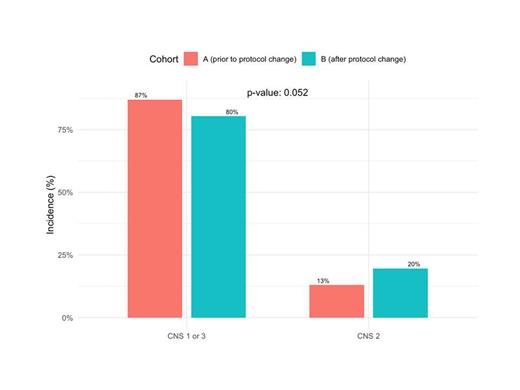
During the study period, 391 patients were diagnosed with B-ALL at CHLA. These included 192 patients prior to the CNS status-based therapeutic change (cohort A) and 199 patients after the therapeutic change (cohort B). The incidence of CNS2 status was higher in cohort B (20%) than in cohort A (13%) (p=0.052). Conversely, the incidence of CNS1 status was lower in cohort B (77%) than in cohort A (85%), while the incidence of CNS3 was similar before (5%) and after (4%) the protocol change. There were no differences between the cohorts in patient age at diagnosis [cohort A: mean age 8.91 years (SD 5.67 years) vs. cohort B: mean age 8.10 (SD 5.62 years); p=0.20], sex (cohort A: 59.9% male vs. cohort B: 58.8% male; p=0.80), or ethnicity (cohort A: 76.0% Hispanic vs. cohort B: 67.7% Hispanic; p=0.09).
These data suggest that the incidence of CNS2 status in pediatric patients with B-ALL at our institution was higher after (20%) than before (13%) the COG protocol changes that began treating CNS2 patients more intensively than CNS1 patients. In addition to increased frequency of intrathecal chemotherapy, in the ongoing frontline COG standard-risk ALL trial, patients with CNS2 status and neutral cytogenetics are upstaged to higher-risk therapy irrespective of end-induction response. A limitation of this study is that pathologist reviewers of CSF cytospin morphology at the time of CNS assignment differed during the two time periods, such that an observational bias cannot be entirely excluded. Notably, flow cytometry - a more sensitive methodology than cytospin morphology for detection of leukemic cells in CSF material - was not used in these cohorts, but increasingly widespread adoption of such techniques may further increase the incidence of CNS2, including in patients who would otherwise have been diagnosed with CNS1 disease (by cytospin morphology) and as such may be overtreated. Future directions of our study include examination of clinical outcome as well as recent (2021-present) patterns of flow cytometry use in CSF specimens and associated diagnoses of CNS disease in pediatric patients with B-ALL at diagnosis.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal